Metallographic preparation of cast iron

Cast iron is used in many applications, particularly in the automotive and engineering industries, and metallography is an important part of quality control in the production process. Find out how to perform accurate metallographic analysis of cast iron, with reproducible results.
The main characteristics of cast iron
Iron is one of the most diverse metals, and it has been an important construction material for centuries, used in many buildings from train stations and market halls to offices and homes.
When alloyed with carbon and other elements, iron offers an enormous variety. The term cast iron refers to iron-carbon-silicon alloys that contain 2.5-4 % carbon and usually 1-3 % silicon. In grey irons, the carbon is present as graphite; in white cast iron it is present in the form of iron carbide and alloy carbides.
Cast iron has good castability and machinability, as well as moderate mechanical properties. Because of its economic advantages, cast iron is used for many applications in the automotive, manufacturing and processing of metals industries. In addition, specific cast irons are the material of choice for sea water pump housings, rolling mill rollers and parts for earth moving equipment.

Fig. 1: Austempered ductile iron, Beraha color etch (Mag. 500x, DIC)
The metallographic quality control process for cast iron
As the morphology of graphite has a major influence on the mechanical properties of cast iron, metallographic quality control of grey iron is an integral part of the cast iron production process.
The basic metallographic quality control process for cast iron is as follows:
- The morphology, size and distribution of the graphite is determined on an unetched and polished sample, using standard reference comparison charts and/or image analysis.
- Depending on the specification, the sample is then etched to check the structure of the matrix.
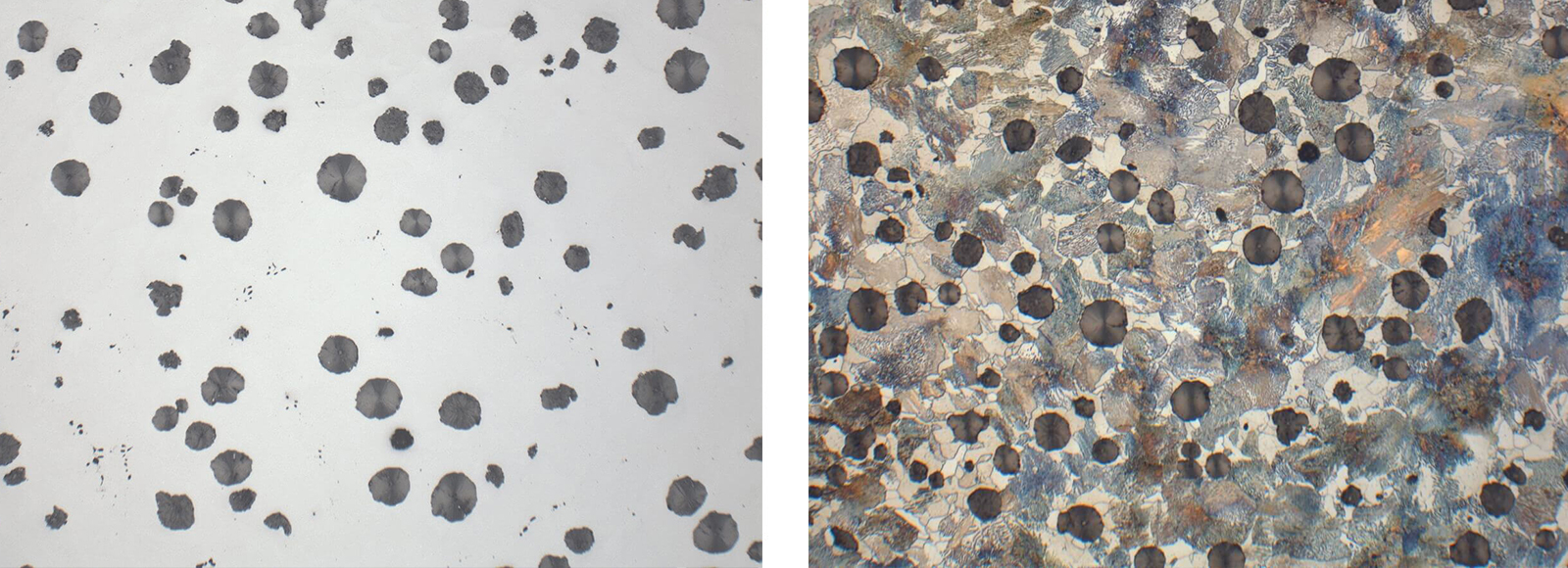
Fig. 2: Cast iron GGG60, unetched (Mag: 200x)
Fig. 3: Cast iron GGG60, etched 3 % Nital (Mag: 200x)
Difficulties in the metallographic preparation of cast iron
The main challenge when preparing samples of cast iron is to retain the graphite in its original shape and size to ensure a correct representation of the cast iron’s microstructures.
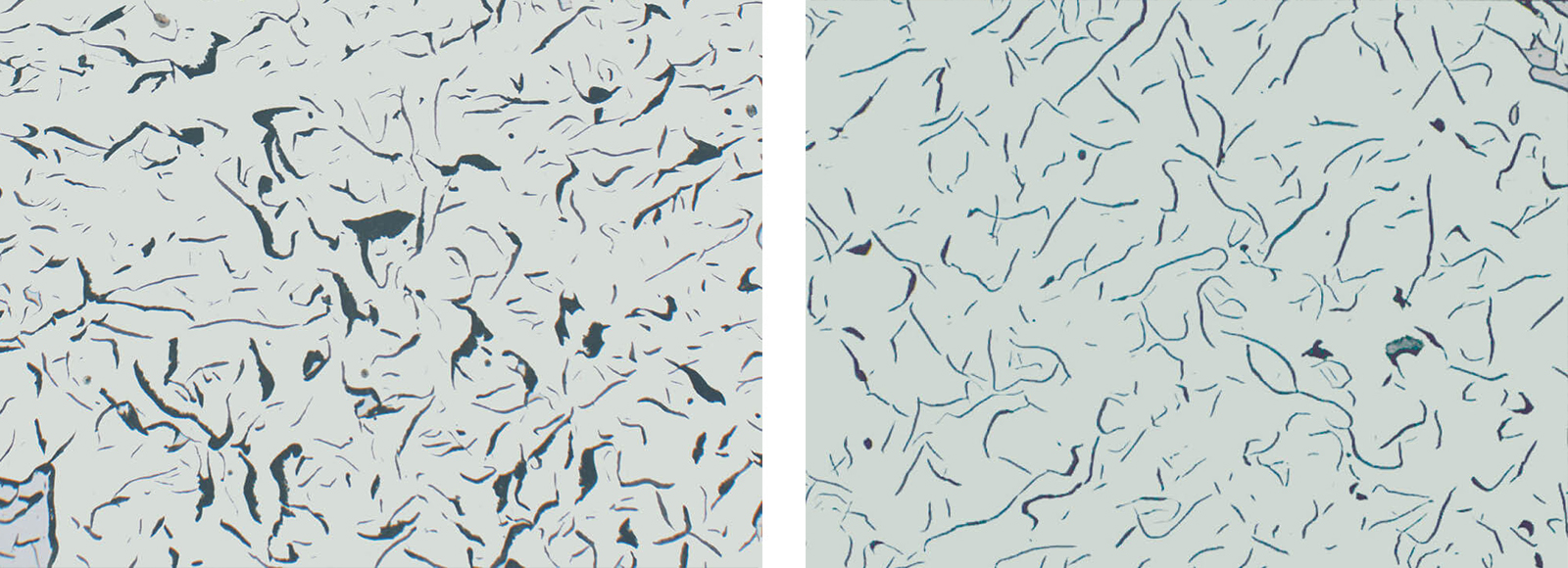
Fig. 4: Grey iron with flake graphite, insufficient polish (Mag: 200x)
Fig. 5: Grey iron with flake graphite, showing correct polish (Mag: 200x)
In the microscope, the image of the graphite is seen two-dimensionally. However, it is actually three-dimensional. This means that a certain percentage of graphite is cut very shallow during grinding and polishing, with only a weak hold in the matrix. Therefore, there is always a possibility that the graphite cannot be completely retained, especially very large flakes or agglomerations of flakes. As a result, the graphite phase cannot always be retained or polished well.
In malleable cast irons, graphite exists in the form of rosettes or temper carbon. This is a friable form of graphite and it can be particularly difficult to retain during metallographic preparation.
A common preparation error is the insufficient removal of smeared matrix metal after grinding, which can obscure the true shape and size of graphite. This is particularly prevalent in ferritic and austenitic cast irons, which are prone to deformation and scratching. For these materials, a thorough diamond and final polish is very important.
Most standard microscopic checks of cast irons are done with a magnification of 100x, which makes the graphite appear black. However, higher magnifications are required to verify if the carbon is completely retained, as well-polished graphite is grey.
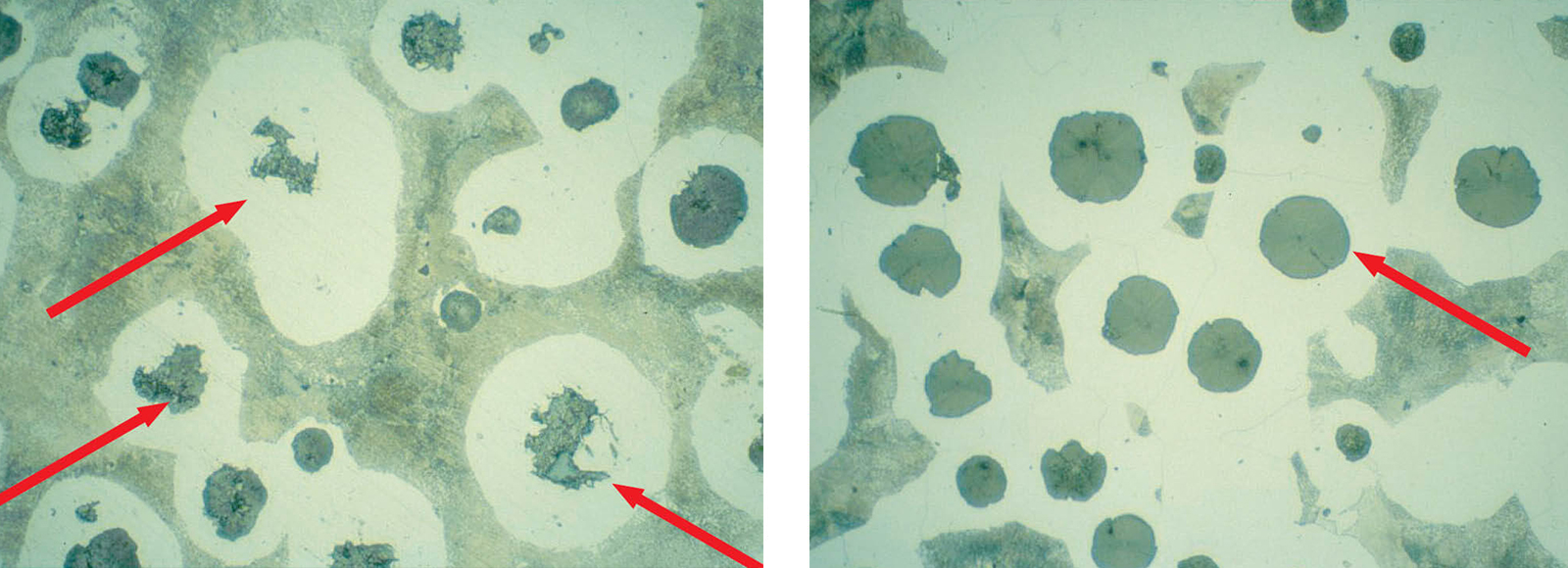
Fig. 6: Insufficient polish leaves graphite nodules covered with smeared metal, etched 3 % Nital (Mag: 200x)
Fig. 7: Correct polish shows shape and size of graphite nodules suitable for evaluation, etched 3 % Nital (Mag: 200x)
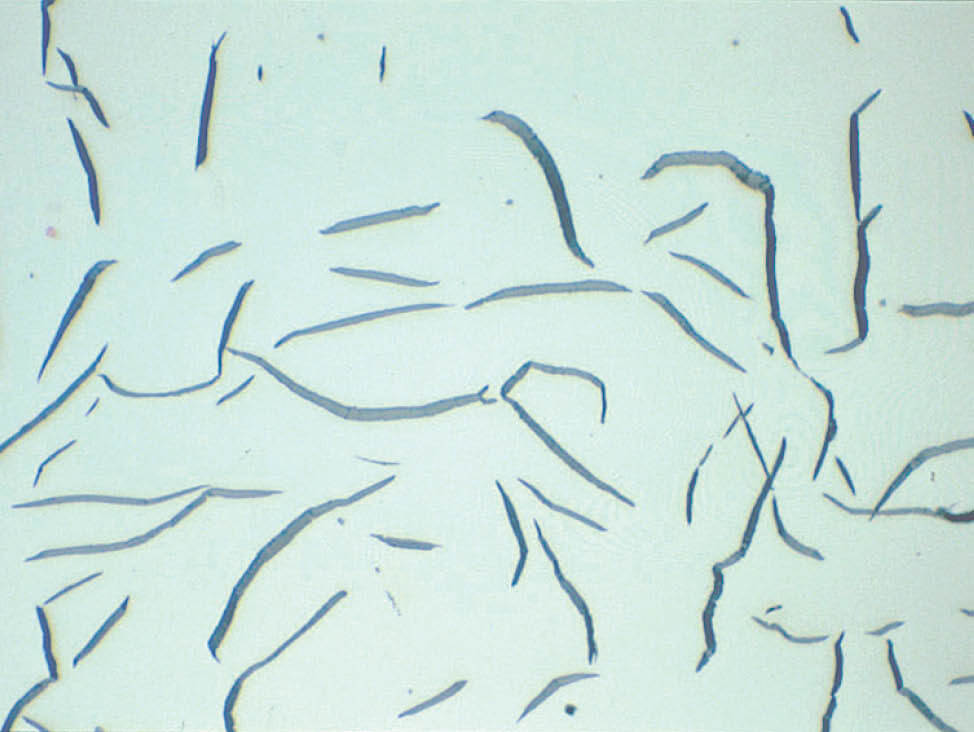
Fig. 8: Well-polished graphite flakes (Mag. 500x)
Overcoming time constraints
Time constraints often make it difficult to maintain consistent preparation results when using manual methods for the metallographic preparation and microstructure analysis of cast iron. However, due to the geometry of the test piece, automatic preparation is often not possible. The solution is to design specific test pieces to fit into an automatic system.
Recommendations for the cutting and mounting of cast iron
Cutting
- Alloyed white cast irons are very hard (HV 600) and can be difficult to cut, especially large sections. Despite this, diamond cut-off wheels are not suitable for cutting white cast iron. Instead, a cubic boron nitride wheel is recommended.
- For large sections, automatic cutting is more efficient than manual cutting.
- For cutting cast irons with graphite, it is recommended to select an aluminum oxide wheel according to the hardness of the cast iron.
Mounting
- Quality control samples are usually prepared unmounted.
- For failure analysis samples, it is best to use hot compression mounting.
- For soft to medium-hard cast irons, a phenolic resin (MultiFast) is recommended – for optimum edge retention use LevoFast (melamin resin with mineral and glass filler).
- For harder cast irons, a reinforced resin (DuroFast) is suitable.
Find out more
- Find more knowledge, expertise and insight in our cutting and mounting sections.
- See our range of cutting and mounting equipment.
- Find cutting consumables and accessories and mounting consumables and accessories.
Recommendations for grinding and polishing of cast iron
See below to see the full step-by-step preparation method for white cast irons and cast irons with graphite.
- Diamond on rigid discs is recommended for fine grinding of most cast irons, as it keeps the samples very flat and does not leave the graphite in relief.
- Hard white cast irons and austempered ductile irons can be plane ground with diamond (MD-Piano 220) and fine ground with diamond on MD-Allegro.
- Soft and medium-hard cast irons with a ferritic, austenitic or pearlitic matrix should be plane ground with silicon carbide foil/paper and fine ground with diamond on MD-Largo.
- For cast irons that tend to corrode during polishing, a water-free diamond suspension (DP-A) and yellow lubricant is recommended.
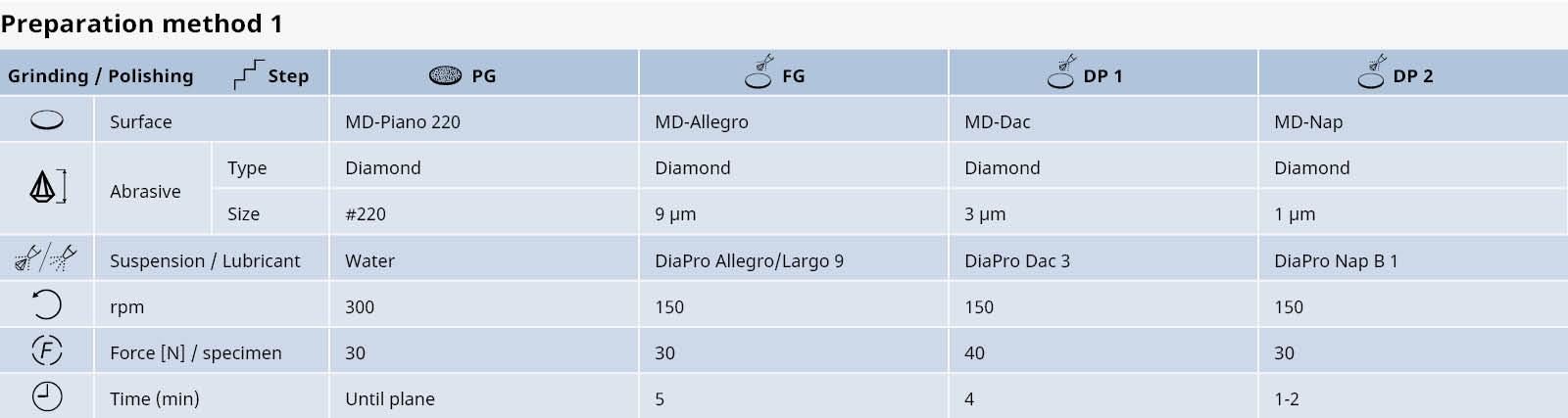
Table 1: Preparation method for white cast irons.
Alternatively DiaPro diamond suspension can be replaced by DP-SuspensionP, 9 μm, 3 μm and 1 μm respectively, applied with DP-Blue lubricant.
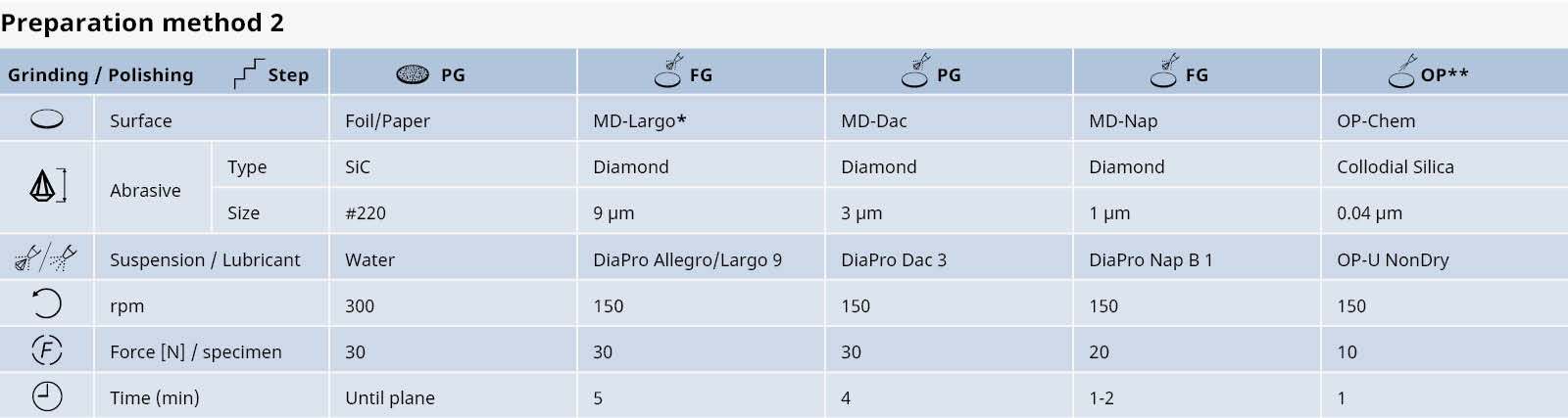
Table 2: Preparation method for cast irons with graphite.
*In cases where retention of graphite is very difficult, MD-Plan cloth can be tried for fine grinding.
**This step is optional
Alternatively DiaPro diamond suspension can be replaced by DP-Suspension P, 9 μm, 3 μm and 1 μm respectively, applied with DP-Blue lubricant

Fig. 9: Grey iron prepared with fine grinding on silicon carbide foil/paper, still showing scratches
Fig. 10: Grey iron, prepared with fine grinding using diamond on rigid disc MD-Largo, showing good edge retention
Find out more
- Find more knowledge, expertise and insight in our grinding and polishing section.
- See our range of grinding and polishing machines and equipment.
- Find consumables and accessories for metallographic grinding and polishing.
Recommendations for cleaning of cast iron
- As many cast irons tend to corrode, cleaning must be fast and should always be carried out with cold water. However, the sample must not be left in contact with water.
- Thorough rinsing with ethanol and fast drying with a strong stream of warm air is recommended. If corrosion still occurs, clean and rinse with water-free alcohol only.
Recommendations for etching of cast iron
- Cast iron samples are initially microscopically examined unetched to evaluate the shape, size and distribution of graphite, as well as cast porosity. After this initial evaluation of the microstructure, the sample can be etched for microstructure with 1-3 % Nital.
- Cast irons with graphite are not suitable for electrolytic polishing as the graphite is washed away by the electrolyte. However, if only a quick identification of the microstructure of the matrix is required, electrolytic polishing and etching can be used.
The following Beraha reagent can be used for color etching and can be modified according to the alloy:
1000 ml water
200 ml hydrochloric acid
24 g ammonium difluoride
To 100 ml of this stock solution, add 1 g potassium metabisulfite
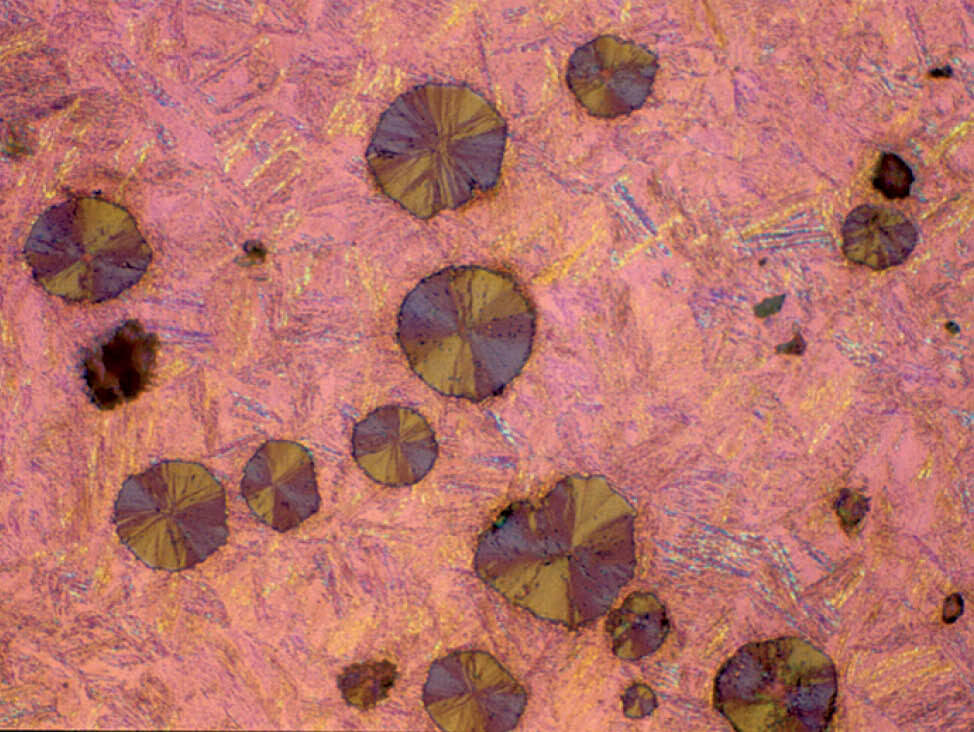
Fig. 11: Austempered ductile iron, etched with 3 % Nital, pol. light (Mag: 200x)
Download the application note to see the full preparation method
Summary
Cast irons are ferrous alloys with mostly 2.5-4 % carbon and 1-3 % silicon. The main difficulty in the metallographic preparation of cast iron is to retain the true shape and size of the graphite in its flake, nodular or tempered form. In particular, cast irons with a soft ferritic matrix tend to smear and are prone to deformation and scratching.
During grinding, the matrix is smeared over the graphite and, unless it is followed by a very thorough diamond polish, the graphite is not shown in its true form. Therefore, plane grinding with silicon carbide foil/paper is recommended, followed by fine grinding and polishing with diamond. A brief final polish with colloidal silica is optional.
Get insight into other materials
If you would like to learn more about the materialography of other metals and materials, check out our materials page.
Get the full application note
Want to read the full application note on the metallographic preparation of cast iron? Download it here.

All images by Birgitte Nielsen, Application Specialist, Denmark
For specific information about the metallographic preparation of cast iron, contact our application specialists.