Metallographic preparation of aluminum and aluminum alloys
The metallographic challenges associated with aluminum and its alloys vary greatly, depending on the metal’s purity. Here, you can learn how to prepare any aluminum product for metallographic examination quickly and effectively.

The main characteristics of aluminum and its alloys
Aluminum is a multifaceted material with multiple uses, including as a matrix metal for composites. It has a silvery white appearance and is used as either a pure metal or as an alloy.
It is extremely light and just small amounts of alloying elements can increase its strength. It is also highly resistant to corrosion. This is due to a passive film of aluminum oxide that is intimately connected to the surface and capable of renewing itself spontaneously when the surface is damaged.
Aluminum’s other significant properties include its high heat conductivity and easy formability – either by casting, hot or cold working or machining – as well as its neutral taste and non-toxicity.
Common uses of aluminum or its alloys:
- High-strength/low-weight applications in the aircraft, aerospace and automobile industries
- Polished and brushed surfaces, as well as anodized colors, in the building industry
- Non-toxic/taste-free packaging and machinery in the food industry
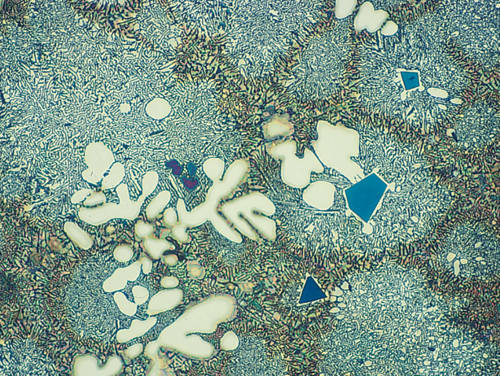
Fig. 0: Aluminum-silicon cast, color etched with molybdic acid, Mag 200x
The production of aluminum
Economical extraction of aluminum is only possible from bauxite. The production process involves two basic steps.
Extraction of pure alumina
Alumina recovery begins by crushing and finely grinding the bauxite and heating it with sodium hydroxide under pressure. In this process, a water-soluble sodium aluminate is formed together with undissolved residues of iron, titanium and silicon. ‘Seed crystals’ of fresh aluminum hydroxide are added to initiate the precipitation of pure aluminum hydroxide (Al(OH)3). Through calcination at 1200 °C, the water is then removed and pure anhydrous alumina (aluminum oxide) remains.
Converting alumina to aluminum (the Hall-Heroult process)
The reaction chemistry of pure alumina requires an electro-chemical process to extract aluminum from its oxide. As the melting point of aluminum oxide is very high (2050 °C), it is mixed with cryolite to reduce the melting point.
Electrolysis takes place in a large carbon or graphite lined steel container that contains steel rods for conducting electricity and carbon blocks as anodes. During electrolysis, the carbon of the anode reacts with the oxygen of the alumina and, in a secondary reaction, metallic aluminum is produced with the formation of carbon dioxide: 2Al2O3 + 3C → 4Al + 3CO2.
This process produces aluminum of 99-99.9 % purity. Much of this is used for aluminum alloys.
Aluminum alloys
Adding very small amounts of alloying elements to aluminum can increase tensile strength, yield strength and hardness compared to pure aluminum. The most important alloying elements are Si, Mg, Cu, Zn and Mn. These mostly eutectic compounds must be finely dispersed through a hot working process before the alloy can be cold worked.
Ageing of aluminum alloys
Many aluminum alloys are age hardened to improve the mechanical properties. This can be done either naturally or artificially.
- Natural age hardening (example AlCuMg). After solution annealing, the workpiece is quenched and consequently the precipitation of the Al2Cu in the solid solution is sup- pressed. The workpiece is then left to age in ambient temperature. During this process the aluminum lattice precipitates the copper from the supersaturated solution. The resultant strain produced in the aluminum lattice leads to an increase in strength and hardness. The process takes 5-8 days.
- In artificial age hardening, ageing takes place at an elevated temperature, which reduces process time. With an AlMgSi alloy, for example, ageing occurs in 4-48 hours at 120-175 °C after solution annealing and quenching. The precipitation of the Mg2Si phase produces internal strain in the aluminum lattice, which results in an increase in strength and hardness.
Wrought aluminum alloys
The main alloying elements for wrought aluminum alloys are copper, magnesium, zinc and manganese. Silicon and iron affect the mechanical properties and corrosion resistance and can either be impurities or alloying elements, depending on the requested purity and application.
Common uses of wrought aluminum alloys:
- Plates in mechanical engineering and mold construction for rolled products, such as sheets and strips, as well as plated products like radiators and heat exchangers
- Plated sheets for specific semi-finished products for aircraft construction or for decorative applications such as trim and reflectors
- Mechanical engineering, conveying and electro technical applications, as well as high-strength sports and leisure products, such as snowboard bindings and mountain bike gears
- Fiber-reinforced aluminum in the aircraft and aerospace industries

Fig. 1: Aluminum alloy 2024, cast, showing eutectic precipitation on grain boundaries, unetched, 200x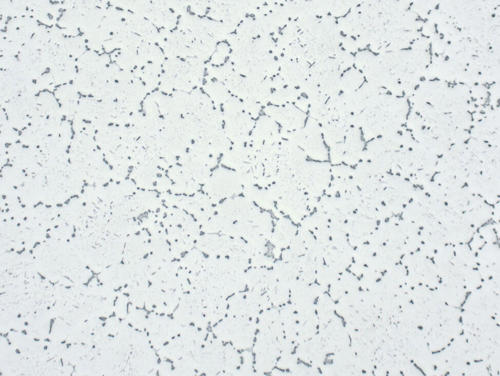
Fig. 2: As Fig. 1, homogenized, unetched, 200x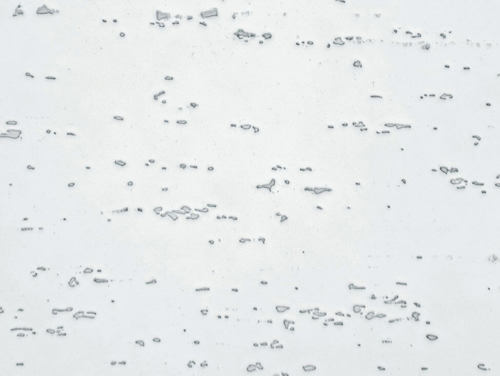
Fig. 3: As Fig. 2, hot rolled, unetched, 200x
Cast aluminum alloys
Aluminum casts are mainly alloyed to improve the metal’s mechanical properties and are differentiated according to their main alloying elements – silicon, magnesium and copper. Alloy contents that exceed the saturation of the solid solution are precipitated as pure metal, such as silicon, or as eutectics and inter-metallic phases.
Silicon increases the castability of aluminum. In eutectic alloys, such as AlSi12, small amounts of sodium are added before casting to refine the eutectic. In this refining process, instead of precipitating as coarse needles or plates (Fig. 4), the silicon forms a very fine eutectic with the α-solid solution (Fig. 5). The effect of hardening in these alloys is very low and therefore magnesium is added so that they can be age hardened.
Cast alloys with specific properties are used in various product groups, including the fabrication of pistons, slide bearings, parts for mechanical engineering, cylinder heads and brake shoes.
| Some of the more important cast alloys and their properties | |
| AlSi10Mg | Age hardened. Vibration and corrosion resistant |
| AlSi5Cu1 | Age hardened. Good castability for welding and thin sections |
| AlMg3 | Resistant to seawater |
| AlSi25+ CuNi | Age hardened. Special alloy for pistons; wear resistant due to its high Si content |
| AlMgSiPb | Suitable for machining |
| AlSi9Cu3 | Castable universal alloy and the most important alloy for pressure die casting |
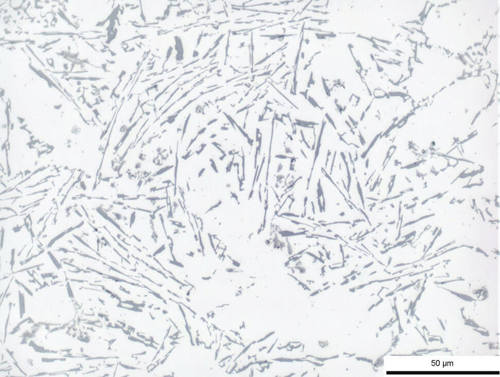
Fig. 4: Aluminum-silicon cast, unrefined, 500x
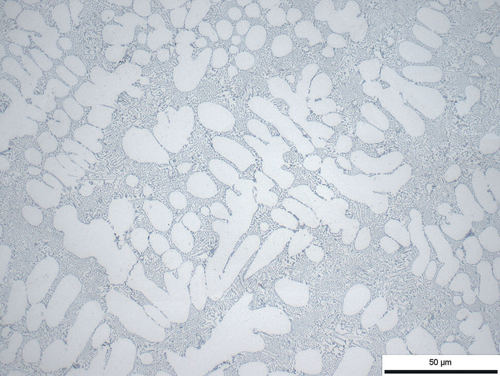
Fig. 5: Aluminum-silicon cast, refined, 500x
The metallography of aluminum and its alloys
Metallography of aluminum is used in quality control for grain size determination and to determine microstructure defects on polished and etched specimens. In addition, specimens are often checked for impurities, such as oxides or zirconium aluminides.
Cast alloy aluminum is evaluated for shape, distribution of phases and possible porosity. In wrought material, defects from the rolling and extrusion process are investigated and plating thicknesses measured.

Fig. 6: Oxide in the surface of an aluminum pressure die casting, 50x
Challenges in the metallography of aluminum and its alloys
The metallographic challenges associated with aluminum and aluminum alloys change with the metal’s purity.
- As purity increases, aluminum becomes softer and more susceptible to mechanical deformation and scratches. In high purity aluminum, grinding can cause deep deformation, while grinding and polishing abrasives, such as silicon carbide and diamond particles, can be pressed into the surface.
- As alloying content increases, aluminum becomes harder. Cast alloys are relatively easy to prepare. However, the aluminum matrix must be well polished to avoid errors in structure interpretation.
| Overview of metallographic challenges and solutions | |
| Challenge: | Solution: |
| Pure aluminum is very soft and prone to mechanical deformation and scratching | Plane grinding with the finest possible SiC Foil or Papers |
| Silicon carbide and diamond particles can be pressed into the specimen surface | Diamond polishing and/or final polishing need to be long enough to remove all embedded particles |
| Severely worked and deformed wrought alloys are difficult to contrast, making structure interpretation difficult | - Final polishing with colloidal silica suspension - Anodization with Barker’s reagent |
Read further for a detailed method description of how to prepare aluminum and its alloys for metallographic analysis quickly and accurately.
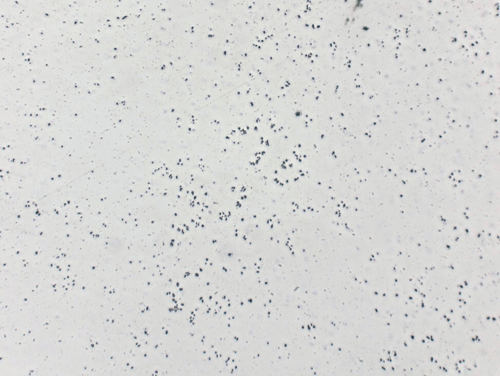
Fig.7: Embedded diamond particles in pure aluminum after polishing with 3 μm, 200x
Preparation of aluminum and its alloys: Cutting & mounting
The cutting and mounting of aluminum and aluminum alloys are simple.
- When sectioning aluminum, use a hard silicon carbide cut-off wheel that is suitable for non-ferrous metals.
- When mounting, a phenolic resin is usually sufficient. However, thin sheets/foils and specimens for anodizing should be mounted in slow-curing epoxy resins, with the specimen protruding from the back of the mount to serve as electrical contacts.
Find out more
- Get more knowledge, expertise and insight on our cutting and mountingsections
- See our range ofCutting equipmentand consumables
- See our range of Mounting quipmentand consumables
Preparation of aluminum and its alloys: Mechanical grinding & diamond polishing
When working with aluminum and its alloys, we recommend mechanical grinding, followed by diamond polishing. For many pure aluminum and wrought alloy specimens, electrolytical polishing is also recommended.
Mechanical grinding
Plane grinding should be carried out with the finest possible grit to avoid excessive mechanical deformation.
- The hardness, size and number of specimens should be considered. However, even with large specimens of pure aluminum, plane grinding with 500# SiC Foil or Paper is usually sufficient.
- Large cast parts of aluminum alloys can be ground with 220# SiC or 320# SiC Foil. It is important that the grinding force is low to avoid deep deformation and to reduce friction between the grinding SiC Foil or Paper and specimen’s surface.
Diamond polishing
Diamond polishing should be carried out until all deep scratches from grinding have been removed. If water soluble constituents must be identified, we recommend polishing with water-free diamond suspension and lubricant.
Final polish for pure aluminum and aluminum alloys: The polish/check sequence
- Begin polishing. After 1 minute of polishing with OP-U suspension, check the specimen under the microscope.
- If necessary, continue polishing for another minute and check the specimen again.
- Continue this polish/check sequence until the required quality has been achieved.
- If diamond particles have been pressed into the surface during polishing, they can lead to erroneous interpretations of the structure. Therefore, the polish/check sequence may need to be relatively long. Continue the sequence until you can no longer see bright and dull areas on the surface of the specimen with the naked eye.
- Approximately 30 seconds before the end of polishing, pour water onto the polishing cloth to rinse the specimen and cloth.
- Finally, wash the specimen again with clean water and then dry it.
Note: Polishing for too long with silicon dioxide suspension OP-S NonDry can cause a pronounced relief, see Fig.11.
![]()
*Alternatively, MD-Dac
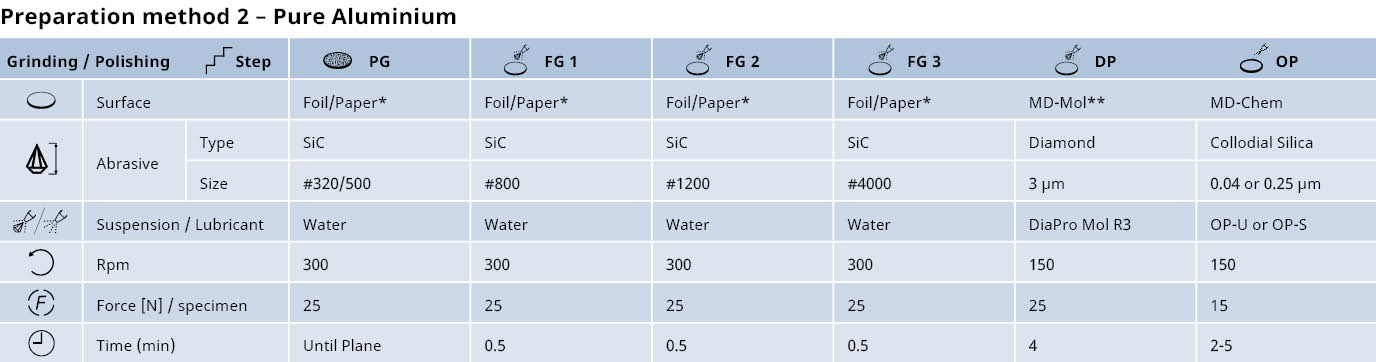
* To avoid coarse scratches, the SiC Foil or Paper can be rubbed with wax before grinding.
** Alternatively, MD-Dac
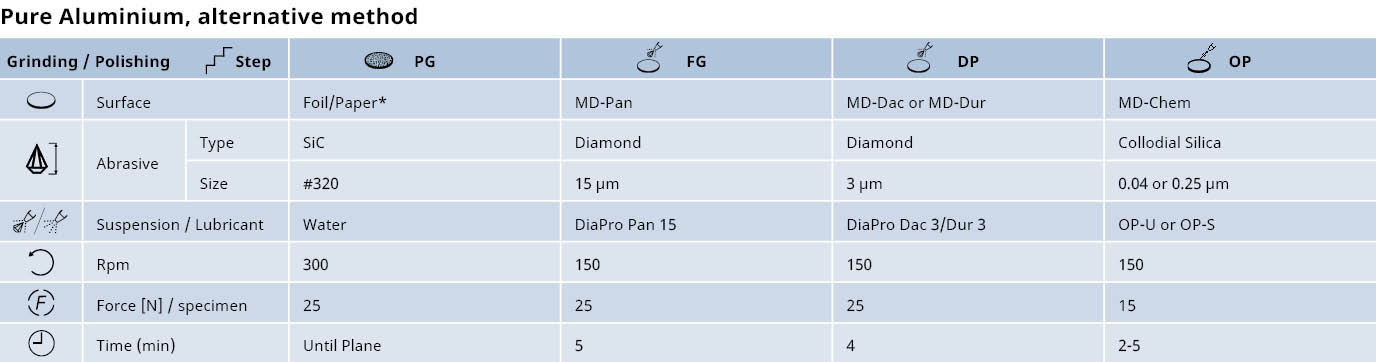
* To avoid coarse scratches, the SiC Foil or Paper can be rubbed with wax before grinding.'

Fig. 9: Aluminum-silicon cast, after polishing with 3 μm diamond small scratches are still visible, 200x

Fig.10: Structure as in Fig. 9, but fine polished with OP-U suspension. The matrix is well polished, and the eutectic has more contrast, 200x
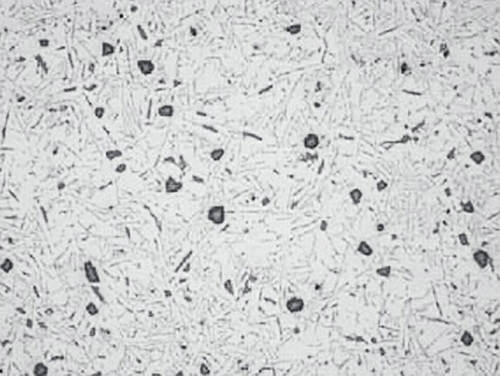
Fig.11: Aluminum-silicon cast polished for too long with OP-S suspension, silicon precipitates stand in relief, 100x
Electrolytic polishing
Electrolytic polishing of aluminum leaves a scratch-free surface and is often used in quality control as it delivers fast and reproducible results. However, it is not recommended for many cast alloy specimens, due to the many different phases in cast alloys.
Pure aluminum and wrought alloys
Electrolytic polishing is especially suitable for pure aluminum and wrought alloys.
- For routine grain size determination at 100x, pre-grinding with 1000# SiC Foil is sufficient.
- Rolled or drawn surfaces do not need any grinding or polishing.
- For pure aluminum and precise examinations of grain shapes, the specimen should be fine ground to 2000# and sometimes even to 4000# before electrolytic polishing.
If you anodize the specimen with Barker’s reagent after the polishing, it will result in a color contrast that is particularly suited for grain size evaluation. To obtain the color effect, view the specimen under polarized light with a λ1⁄4 sensitive tint plate.

Fig.12: Plated sheet, anodized, grain areas are clearly visible and suitable for automatic image analysis, polarized light with λ1⁄4-plate, 100x
Discover the parameters for electrolytic polishing of aluminum in our application note here.
- Get more knowledge, expertise and insight on our grinding and polishingsection
- See our range ofgrinding and polihsing equipment and products
- Get consumablesand accessoriesfor metallographic grinding and polishing

Fig.13: Aluminum pressed part, macro etching, primary and heterogeneous precipitates are revealed
Etching of aluminum and its alloys
When working with aluminum and its alloys, macro etchants are used for grain size evaluation, also to show flow lines from extrusion and to reveal weld seams. Before etching, the specimen has to be ground with 1200# SiC Foil or 2400# SiC Foil.
Due to the many alloying possibilities of aluminum, the different phases cannot always be clearly identified in some of the multi-component alloys. However, the eutectic phases can sometimes be recognized by the typical shape of their eutectic.
Some of the well-known phases have the following characteristic colors:
- Si: Grey
- Mg2Si: Tarnished dark blue during polishing (in cast: Chinese script)
- Al2Cu: Pinkish-brown, copper colored
- Al6Mn: Light grey
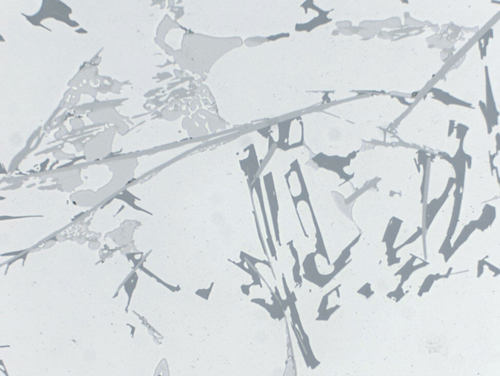
Fig. 14: AlSi6Cu4 cast, the various phases can be identified by their typical color, 500x
Manual grain size determination in wrought alloys
Certain specifications for wrought alloys require ‘manual’ grain size determination, as the image analysis is not precise enough. In addition, wrought alloys with a heavily deformed structure, such as foils or very thin sheets, are difficult to contrast with chemical etchants. This can make automatic image analysis difficult.
It is recommended to anodize the specimen with Barker’s reagent and then evaluate the structure ‘manually’ under the microscope.
Etching solutions
When working with chemicals the standard safety precautions must be observed.
| Macro etching | |
| For pure aluminum | 90 ml water |
| 15 ml hydrochloric acid | |
| 10 ml hydrofluoric acid | |
| Deep etching to reveal the primary dendritic structure | |
| 100 ml water | |
| 10-25 g sodium hydroxide |
| Micro etching | |
| Flick’s reagent:Grain boundary etching for most types of aluminum and alloys | |
| 90-100 ml water | |
| 0.1-10 ml hydrofluoric acid | |
| Dix and Keller reagent:Grain area etching for Al-alloys with copper, also suitable for pure aluminum | |
| 190 ml water | |
| 5 ml nitric acid | |
| 10 ml hydrochloric acid | |
| 2 ml hydrofluoric acid | |
| Color etchants: | Molybdic acid solution according Klemm or Weck |

Fig.15: Experimental aluminum alloy with 6% Si and 10% copper, unetched

Fig.16: Same as Fig.15 but etched for 30 seconds with 1 g molybdic acid in 200 ml water + 6 g ammonium chloride. The silicon is dark blue and is distinguishable from the greyish CuAl2
Download the application note to see the full preparation method
Summary
Its low density, high strength and corrosion resistance make aluminum and its alloys the material of choice for many applications in the automobile, aircraft, aerospace, packaging and other industries.
Metallography of aluminum and its alloys is used in quality control for grain size determination, and the evaluation of phases, impurities and mechanical defects.
Pure aluminum is very susceptible to deformation and therefore grinding should not be carried out with coarse grits. A very thorough final polishing with silicon dioxide suspension is necessary to ensure that embedded diamond particles are completely removed from the specimen’s surface.
Aluminum cast alloys are polished relatively easily. For grain size evaluation, anodizing with Barker’s reagent will result in a better contrast than chemical etching. Different phases in cast alloys can either be identified by their characteristic color or by etching with specific solutions that attack certain phases preferentially.
Get the full application note
Want to read the full application note on the metallographic preparation of aluminum and its alloys?Download it here
Get insight into other materials
If you would like to learn more about the materialography of other metals and materials, check outour materials pages.

All images by Xiuping Jiang, Application and Laboratory Manager, China
For specific information about the metallographic preparation of aluminum, contact our application specialists.