Metallographic preparation of stainless steel

The majority of stainless steels are soft and prone to deformation and scratching during metallographic preparation, which makes an accurate preparation method essential. Find out how to prepare stainless steel for metallographic examination with reproducible results.
The main characteristics of stainless steel
Stainless steels are corrosion resistant steels that contain at least 11 % chromium. Within this group of high alloy steels, four main categories can be identified:
- Ferritic
- Martensitic
- Austenitic
- Austenitic-ferritic (duplex)
These categories describe the microstructure of the stainless steel alloy at room temperature, which is largely influenced by the alloy composition, as well as the heat treatment.
High corrosion resistance
The main characteristic of stainless steels is their corrosion resistance, which can be enhanced by the addition of specific alloying elements. These alloying elements also have a further beneficial effect on other material characteristics, such as toughness and oxidation resistance.
For example, niobium and titanium increase resistance against intergranular corrosion as they absorb the carbon to form carbides; sulfur increases machinability, because it forms small manganese sulfides, which result in short machining chips.
Metallography of stainless steel
Due to their corrosion resistance and superior surface finish, stainless steels play a major part in the aircraft, chemical, medical and food industries, in professional kitchens, architecture and even jewelry. Stainless steels are also commonly used in automotive applications.
Metallography of stainless steels is an important part of the overall quality control process in many production environments. The main metallographic tests are:
- Grain size measurement
- Investigation of general structure, including the content of martensite, ferrite, perlite or austenite
- Identification of delta ferrite and sigma phases
- Evaluation of carbides and their distribution
- Investigation of welds
In addition, metallography is used in failure analysis to investigate corrosion/oxidation mechanisms. 
Fig. 1: Duplex steel etched electrolytically with 40 % aqueous sodium hydroxide solution, showing brown austenite and blue ferrite. Bright field.
Preparation of stainless steel: Grinding & polishing
Ferritic stainless steels are soft; austenitic steels are ductile. Both are prone to mechanical deformation during the metallographic preparation of stainless steel.
Final polishing usually leaves these steels highly reflective. However, if they are not thoroughly pre-polished, deformation can reappear after etching. Due to their hardness, martensitic steels are relatively easy to polish. However, care should be taken to preserve the carbides. 
Fig. 2: Austenitic steel insufficiently polished showing deformation after 3 µm polishing and etching with Beraha III. Bright field.
Overcoming difficulties in grinding & polishing stainless steels
During grinding and polishing, the retention of carbides and inclusions can be a problem. In addition, deformation and scratching can occur in ferritic and austenitic stainless steels.
The solution is to use thorough diamond polishing and complete final polishing with colloidal silica or alumina.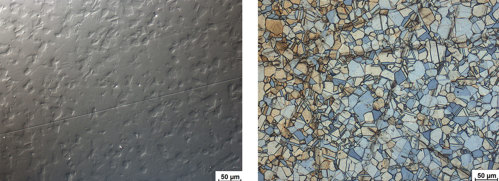
Fig. 3.1: Insufficiently polished stainless steel showing still deformation visible in DIC, not etched
Fig. 3.2: Surface of stainless steel after final polishing, showing deformation from grinding or fine grinding. Etched with Beraha III. Bright field
Electrolytic polishing of stainless steels
For research work or a quick general microstructure check of stainless steel, electrolytical polishing and etching can be an alternative to mechanical polishing, as it does not leave any mechanical deformation.
Electrolytic polishing gives excellent results for checking the microstructure of stainless steel. However, the process can dissolve some elements, such as small carbides, and so care should be taken.
Before electrolytic polishing, the samples have to be ground to 500# or finer on silicon carbide foil/paper. The finer the initial surface, the better the results of the electrolytical polish.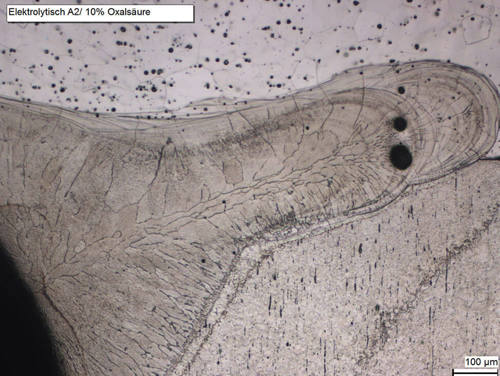
Fig. 4: Laser weld in stainless steel, unmounted, electrolytically polished with A2 and etched with oxalic acid in water 10 %. Bright field.
Preparation method
Electrolytic polishing and etching of stainless steel (grinding on SiC foil/paper 220#, 500# and 1000#):
Electrolyte: A2
Area: 5 cm2
Voltage: 35 V
Flowrate: 13
Time: 25 sec
External etching with stainless steel etching dish:
10 % aqueous oxalic acid
Voltage: 15 V
Time: 60 sec
Recommendations for grinding and polishing of stainless steels
Grinding
- For soft and ductile stainless steels, it is strongly recommended that the use of very coarse grinding foil/papers and high pressures are avoided, as these can result in deep deformation.
- As a general rule, the finest possible grit, consistent with the sample area and surface roughness, should be used for plane grinding.
Polishing
- Any deformation from the first grinding step that is not removed by fine grinding will leave its traces. These traces can be removed by final polishing, but it is time-consuming.
- Fine grinding should be carried out with diamond on a rigid disc (MD-Largo) or (as an alternative for some types of stainless steels) on a MD-Plan or MD-Sat cloth.
- Fine grinding should be followed by a thorough diamond polish on a medium hard cloth, before a final polish with colloidal silica (e.g. OP-S) or alumina (OP-A) to remove any fine scratches. This final step should be very thorough and can take several minutes. A good final polish increases the chance of better contrast.
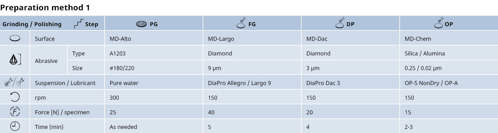
Preparation method for stainless steel samples, 30 mm diameter mounted, on the semi-automated Tegramin, 300 mm diameter. As an alternative to DiaPro, polycrystalline P can be used together with green/blue lubricant.
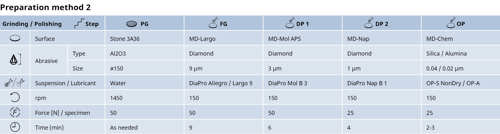
Preparation method for stainless steel samples 65x30 mm cold mounted or unmounted using Struers AbraPlan / AbraPol, 350 mm diameter with stone grinding. As an alternative to DiaPro, polycrystalline P can be used together with green/blue lubricant.
Find out more
- Get more knowledge, expertise and insight on our grinding and polishing section
- See our range of grinding and polishing machines and equipment
- Get consumables and accessories for metallographic grinding and polishing
Etching of stainless steels
Etching stainless steels requires some experience and patience. The literature for etchants is extensive, and we recommend that you try a variety of etchants in order to establish an individual stock of solutions that are suited to the particular materials regularly prepared in your laboratory.
For good etching results, a sufficient final oxide polishing is important. You can learn more about how to use etching to better visualize the microstructure or macrostructure of your sample here.
The following are some etchants that have proved successful in everyday, routine applications.
Chemical etching
Caution: Always follow the recommended safety precautions when working with chemical reagents.
For martensitic steels
1) V2A etchant:
100 ml water
100 ml hydrochloric acid
10 ml nitric acid
Etch at room temperature or up to 50 °C
2)
33 ml water
33 ml ethanol
33 ml hydrochloric acid
1.5 g copper-II-chloride (Kalling I)
3)
100 ml water
100 ml hydrochloric acid
5g copper-II-chloride (Kalling II)
For austenitic steels
1) V2A etchant:
100 ml water
100 ml hydrochloric acid
10 ml nitric acid
Etch at room temperature or up to 50 °C
2) Adler etchant:
25 ml distilled water
50 ml hydrochloric acid
15 g iron-III-chloride
3 g diammoniumtetrachlorocuprat (II)
Can be used for stainless steels undiluted or mixed with water or ethanol up to 1:1
3)
100 ml water
300 ml hydrochloric acid
15 ml hydrogen peroxide (30 %)
Color etchant Beraha II:
Stock solution
800 ml distilled water
400 ml hydrochloric acid
48 g ammonium biflouride
For etching, add 1-2 g potassium metabisulfite to 100 ml of this stock solution
Color etchant Lichtenegger & Bloech:
100 ml distilled water
20 g ammonium bifluoride
0.5 g potassium metabisulfite
Can be used for austenitic and duplex steels
Electrolytic etching
For austenitic-ferritic steels (duplex):
20-40 % aqueous sodium hydroxide solution
All stainless steels:
10 % aqueous oxalic acid
For automatic grain size measurements of austenitic steels:
60 % distilled water
40 % nitric acid
Fig. 5: Austenitic steel, electrolytically etched with nitric acid in water, showing only grain boundairies and no twins. Ideal for automatic grain size measurements
Interpreting microstructures of stainless steel
Ferritic stainless steels do not respond to hardening. Their properties, however, can be influenced by cold working. They are magnetic at room temperature. The microstructure in the annealed condition consists of ferrite grains in which fine carbides are embedded. Ferritic steels used for machining contain a large amount of manganese sulfides to facilitate free cutting.
Martensitic stainless steels respond to heat treatment. Martensite is formed through rapid cooling. Properties can then be optimized by subsequent tempering treatment. The alloys are magnetic. Depending on the thermal treatment, the microstructure can range from a pure martensitic structure to fine-tempered martensite. Different alloys and various dimensions of semi-finished products require complex heat treatment temperatures and times.
Fig. 6: Martensitic chrome steel, electrolytically polished and etched with A2. Bright field.
In some corrosion resistant steel welds, a certain amount of delta ferrite is needed to improve hot-crack resistance. However, delta ferrite is usually an unwanted phase, because the long annealing times of steel with a high chromium content can change the delta ferrite into the hard and brittle iron-chromium intermetallic sigma phase. Heating up to 1,050 °C and subsequent quenching removes the sigma phase and with it the embrittlement.
Austenitic stainless steels do not respond to thermal treatment. Instead, rapid cooling results in the production of their softest condition. In this state, they are non-magnetic and their properties are influenced by cold working. The microstructure of these steels consists of austenite grains, which may exhibit twinning.
Fig. 7: Austenitic steel with twins and segeragtions. Colour etched with Lichtenegger and Bloech. DIC.
Fig. 8: Deltaferrite in an austenitic steel weld (small dark strings) and larger deltaferrite lines in weld part (blue-grey); electrolytically etched with 40 % aqueous sodium hydroxide solution. Bright field
Exposing these steels to elevated temperatures in the region of 600-700 °C can result in the formation of complex carbides within the austenite grains. This leads to an impoverishment of chromium in the austenite solid solution, which increases the susceptibility to intergranular corrosion or oxidation.
Fig. 9: Austenitc steel tube with twins and deformation from cold working; etched with 10 % oxalic acid, DIC
By reducing the carbon to below 0.015 % and adding small amounts of titanium, niobium or tantalum, the risk of intergranular corrosion is reduced, as these elements form carbides in preference to the chrome. Delta ferrite can appear due to critical heat treatment conditions in martensitic steels or cold working of austenitic steels. 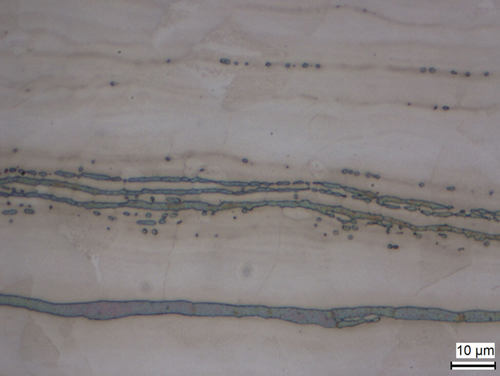
Fig. 10: Strings of deltaferrite in austenitic steel matrix, electrolytically etched with sodium hydroxide in water (20 %)
Austenitic-ferritic stainless steels (duplex) consist of ferrite and austenite. Electrolytic etching in a 20…40 % caustic soda solution reveals the structure, and the correct percentage of each phase can be estimated. These steels are ductile and are specifically used in the food, paper and petroleum industries.
Fig. 11: Forged duplex steel showing blue ferrite, light to dark brown austenite. Double electrolytical etching; first 10 % oxalic acid in water and second etch 20 % sodium hydroxide in water; DIC
Summary
Stainless steels are corrosion resistant steels with high chromium and nickel contents. Ferritic and austenitic steels are softer than martensitic stainless steels and have higher ductility. Hardness and ductility can vary depending on the alloy and the heat treatment. Nevertheless, both types tend to mechanical deformation and scratching during mechanical metallographic preparation. Therefore, electrolytic preparation is a good alternative to mechanical grinding and polishing procedures.
For successful preparation of stainless steel with a mechanical polish, it is suggested that:
- Coarse abrasives for plane grinding are avoided.
- Fine grinding and polishing with diamond should be thorough and ensure removal of all deformation from plane grinding.
- A final oxide polish with colloidal silica or alumina should be carried out to provide a deformation-free surface.
A four-step procedure, completed on automatic preparation equipment, gives good and reproducible results.
Stainless steels are difficult to etch chemically, and the recommended etchants are very corrosive, requiring careful handling. Instead, electrolytical polishing and etching is recommended, as this gives a deformation-free surface but does not always retain carbides.
Get insight into other materials
If you would like to learn more about the materialography of other metals and materials, check out our materials page.

All images by Holger Schnarr, application specialist, Germany
For specific information about the metallographic preparation of stainless steel, contact our application specialists.