Metallographic preparation of copper and copper alloys
Copper is commonly used in a wide range of industries. However, its extreme ductility makes it prone to deformation and scratches. On this page, you can learn how to prepare scratch-free copper samples for metallographic examination quickly and effectively.

The main characteristics of copper and its alloys
Copper is a pliable metal that is easy to shape and form. Its attractive color, corrosion resistance and high electrical and thermal conductivity mean it is widely used in the electronics, automotive, building, chemical, and food and beverage industries.
With increasing purity, the electrical and thermal conductivity of copper increases. Pure, or oxygen-free, copper is only used in very specific applications.
Common industrial uses of copper include:
- Cables, switch-gear components, transformers, motor windings and generators in the automotive industry
- Tubes, vessels and heat exchangers in the chemical, and food and beverage industries
- Facades, roofs, potable water pipes and heating installations in the building industry
- Cryogenics and air conditioning
- Semi-conductors, switches and sealing rings in vacuum technology, and electron tubes in the electronics industry
The copper production process
Although metallic copper occurs naturally, it is mainly extracted from sulfide ores in a metallurgical smelting process. There are four main steps.
1. An initial smelting process extracts copper concentrates, resulting in copper matte (75 % Cu).
2. In a converter, air is blown into the liquid matte to oxidize the sulfides, resulting in blister copper (96-98 % CU).
3. Blister copper is refined in an anode furnace, resulting in anode copper (99 % Cu).
4. Impurities (including Ni, Pb, Ag, Pd and Au) are removed through electrolytic refining, resulting in copper cathodes (99.99 % Cu).
A small amount of copper is also produced by hydro-metallurgical process.

Fig. 1: Copper with red copper oxides, dark field, 500x

Fig. 2: Oxygen-free copper, etched with ammonium peroxydisulfate, 100x

Fig. 3: Copper cathode, etched according to Klemm, 100x
Copper alloys
There is a large number of copper alloys, with zinc (brass) and tin (bronze) being the most common.
Brass (copper-zinc alloys)
Brasses are copper alloys with 5-45 % zinc. As copper has a high solubility for zinc, the alloys are very homogenous. With increasing zinc content, the reddish copper color changes to the yellow of brass. The brass also becomes harder and easier to machine.
- Brass with less than 28 % zinc is called red brass and is especially suitable for machining
- Alloys up to 37 % zinc consist of α- solid solution and are suitable for cold forming
- From 38 % zinc and upwards, the alloys display a two-phase α-β microstructure, which is suitable for hot forming
By adding aluminum, manganese, iron, nickel, tin or even small amounts of lead, special types of brass with specific chemical or mechanical properties can be produced.

Fig. 4: α- brass, color etched, 200x
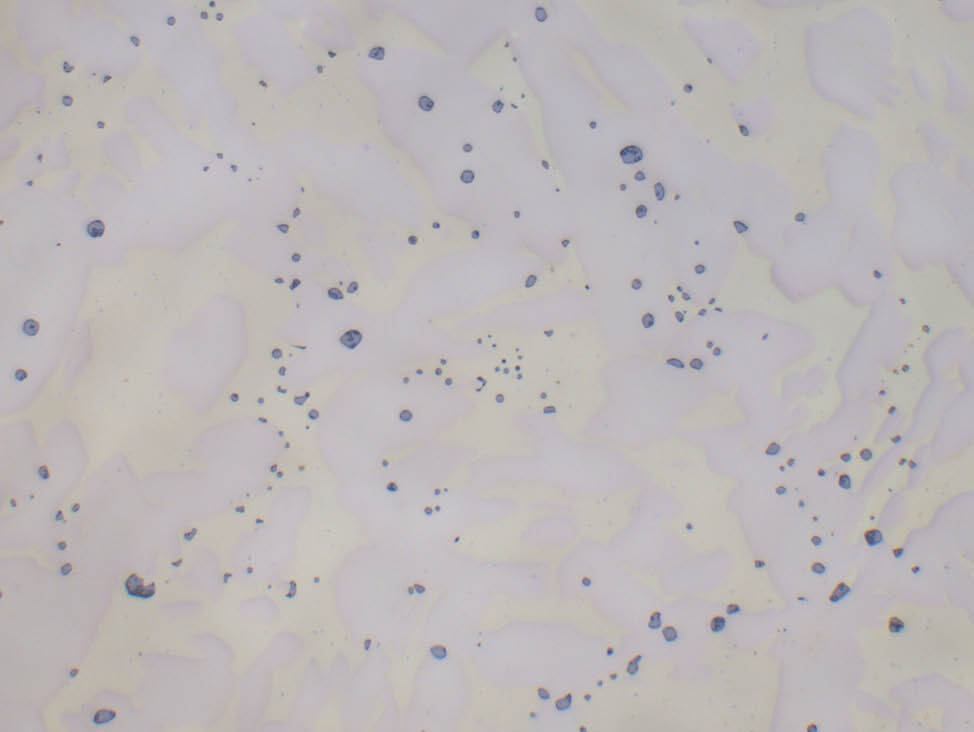
Fig. 5: α-β brass cast (CuZn40Pb2) with grey-blue lead inclusions, unetched, 500x
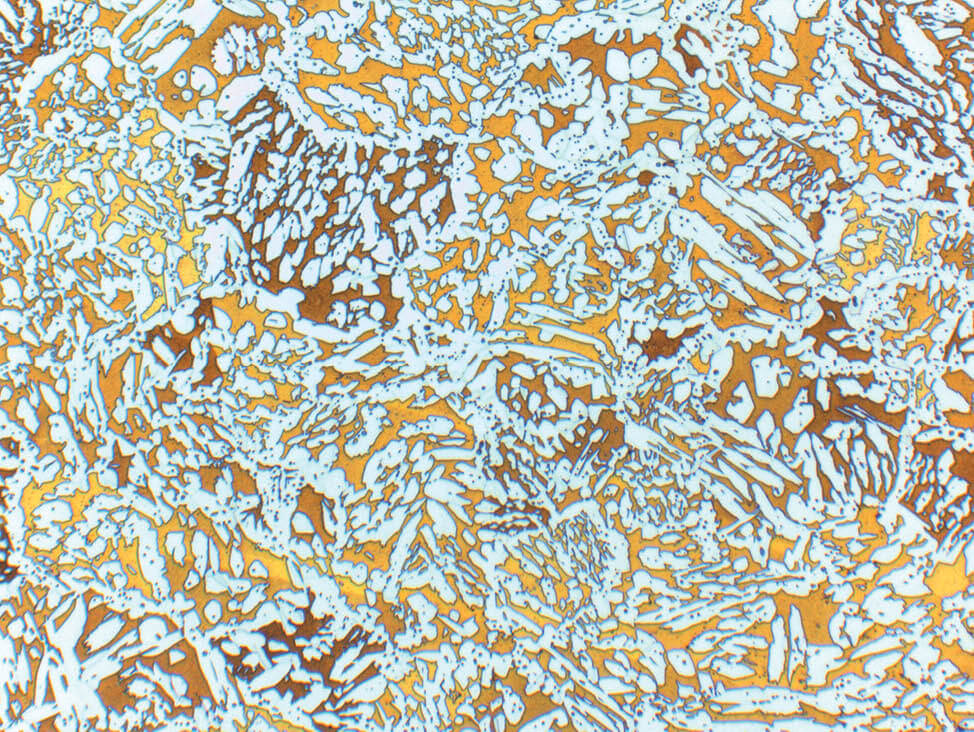
Fig. 6: α-β brass cast, etched according to Klemm, light α- solid solution in dark matrix of β- solid solution, 100x
Bronze (copper-tin alloys)
Bronzes are divided into three types: wrought alloys, cast alloys, and bell casts. Depending on the required properties, small amounts of zinc, phosphorus, lead, nickel or iron can be added.
| Some specific brass alloys and their properties | |
| Gun metal (Cu-Sn-Zn) | Good corrosion resistance; a low friction coefficient |
| Aluminum bronzes (up to 11 % aluminum) | High strength at elevated temperatures; very good corrosion resistance |
| Beryllium bronze | High strength; high hardness; does not produce sparks when striking or impinging other metals |
| Copper-nickel alloys | Excellent corrosion resistance |
| Copper-nickel-zinc alloys (German silver) | High strength; good corrosion resistance; easy to form |

Fig. 7: Aluminum bronze, color etched according to Klemm, pol. light, 200x
The metallography of copper and its alloys
When working with copper and its alloys, metallography is generally used for grain size measurement, as well as to perform purity checks by qualifying and quantifying the copper oxide content.
In certain brasses, it may be necessary to determine the distribution of lead, as this can influence the machining process.
In cast alloys, metallography is generally used for general structure evaluation, to assess the distribution of eutectic or lead, and to evaluate the presence of shrinkage cavities or porosity.
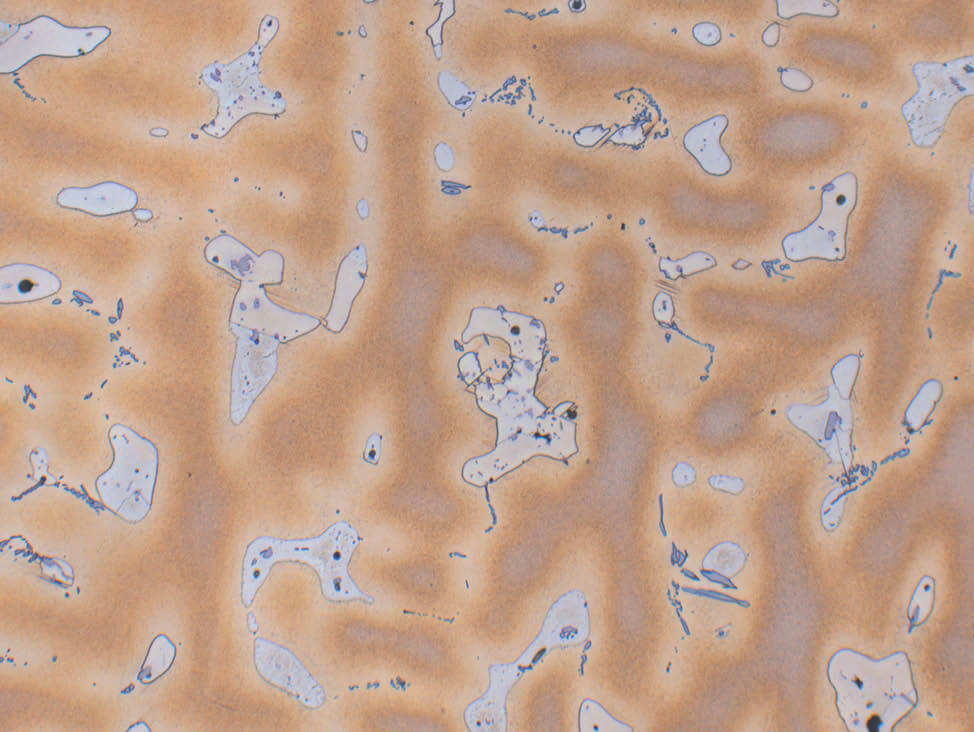
Fig. 8: Bronze cast (CuSn10), etched with iron (III) chloride, dendritic structure α-δ eutectoid, 200x
Challenges in the metallography of copper and its alloys
Pure copper is soft and ductile, which makes it easy to deform and prone to scratches. Even bronzes and some of the harder brasses may be susceptible to severe scratching. This creates a problem for the metallographer. However, there are some simple solutions:
- Avoid coarse grinding abrasives
- Give a thorough diamond polishing with soft cloths
- Use chemical-mechanical fine polishing
Read on for a detailed description of how to prepare copper and its alloys for metallographic analysis quickly and accurately, without scratches or deformation.

Fig. 9: Pure copper wire, with final polish of OP-S, DIC, 200x

Fig. 10: Same sample as Fig. 9 with final polish of OP-S-ammonia/water/hydrogen peroxide mixture, DIC, 200x
Preparation of copper and its alloys: Cutting & mounting
The cutting and mounting of copper and its alloys are fairly simple.
- When sectioning copper or its alloys, use a hard silicon carbide cut-off wheel, suitable for non-ferrous metals
- When mounting copper or its alloys, a phenolic resin will prove sufficient in most cases
Find out more
- Get more knowledge, expertise and insight on our cuttingand mountingsections
- See our range of cutting equipmentand consumables
- See our range of mounting equipmentand consumables
Preparation of copper and its alloys: Mechanical grinding & diamond polishing
With increasing purity, copper becomes softer and more susceptible to mechanical deformation and scratches. Consequently, grinding can cause deep deformation in high purity copper, while grinding and polishing abrasives can be pressed into the surface.
Copper alloys are harder, but still have a tendency to form scratches. In some bronzes, these scratches may occur in some individual grains only.
Note: The preparation data given below are for automatic grinding and polishing of 6 mounted samples, 30 mm in diameter, clamped into a holder.
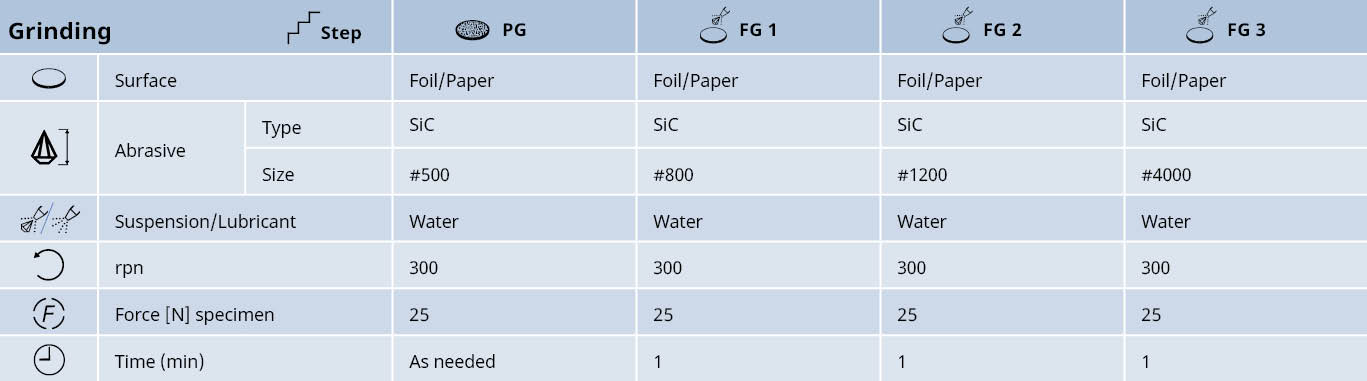
Mechanical grinding
Plane grinding should be carried out with the finest possible grit in order to avoid any excessive mechanical deformation.
- The hardness, size and number of samples should be taken into account. However, even with large samples of pure copper, plane grinding with 500# SiC foil/paper is usually sufficient
- Large cast parts of copper alloys can be ground with 220# or 320#, although it is important that the grinding force is low in order to avoid deep deformation
- Soft alloys should be fine ground using SiC foil/paper with fine grits (up to 4000#)
- For harder alloys, MD-Largo with diamond can be used to ensure better planeness and edge retention
Pure copper and copper alloys with low alloying contents

Diamond polishing
Copper and its alloys should be diamond polished until all deformation and embedded abrasives from mechanical grinding have been removed. Chemical-mechanical fine polishing with silicon dioxide should result in an almost scratch-free surface.
- For pure copper, give a final diamond polish with a solution containing iron nitrate.
- For copper alloys, a mixture of OP-S Non-Dry suspension with hydrogen peroxide and ammonia water is recommended.
Final polish for pure copper and copper alloys: The polish/check sequence
- Begin polishing. After one minute, check the sample under the microscope
- If necessary, continue polishing for another minute and check the sample again
- Continue this sequence until the required quality has been achieved
- If you see that the attack is too fast or too strong, dilute the mixture with water
- Approximately 30 seconds before the end of polishing, pour water onto the polishing cloth to rinse the sample and cloth
- Finally, wash the sample again with clean water and then dry it

Fig. 11: α-β brass cast, mechanically polished, unetched, 200x
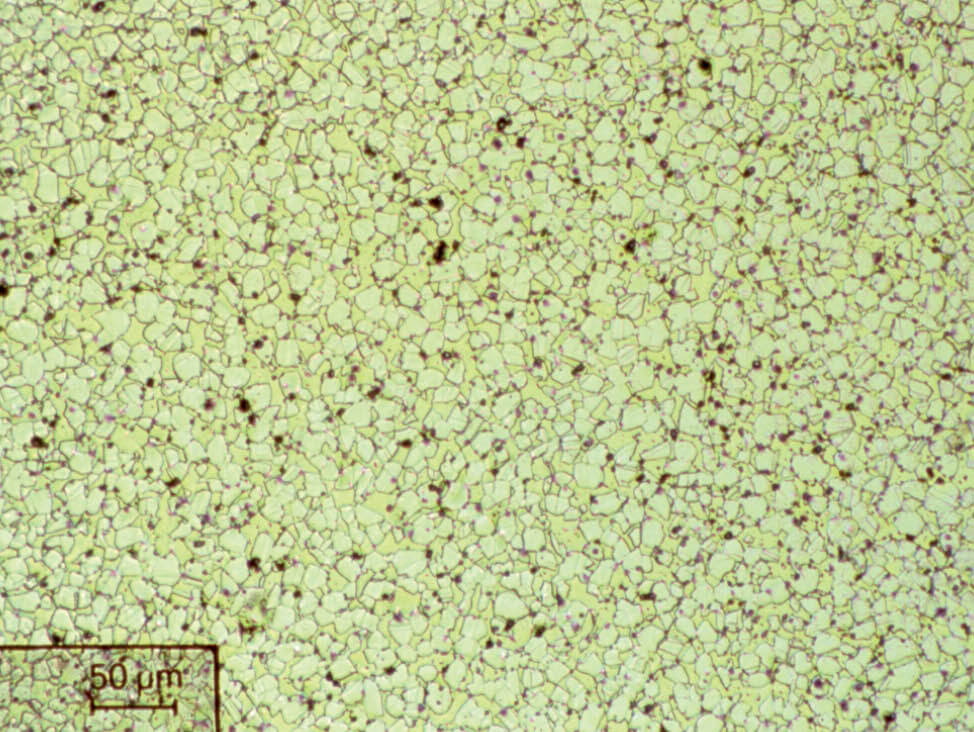
Fig. 12: Same specimen as Fig.11 electrolytically polished, unetched, 200x. The lead inclusions are pulled out and therefore appear larger and more numerous
Copper alloys 
Electrolytic polishing
Electrolytic polishing is suitable for pure copper and α- brass wrought alloys. Two-phase α-β brass can also be polished electrolytically, but the results are not suited to quantitative analysis, especially if the alloy contains lead. Due to the different phases, cast alloys are not suited to electrolytic polishing.
Before electrolytic polishing, fine grinding with SiC foil/paper up to 2400# or 4000# must be performed.
Discover the parameters for electrolytic polishing of copper in our Application Note here
- Get more knowledge, expertise and insight on our grinding and polishing section
- See our range of grinding and polishing equipment and products
- Get consumablesand accessoriesfor metallographic grinding and polishing
Etching of copper and its alloys
There are numerous etchants for copper and its alloys that are relatively easy to apply. Most cast alloys are not difficult to etch. However, it can be difficult to find the right etching solution for some wrought alloys, especially if they have been severely cold worked. In these cases, a color etch can be useful.
It is important to note that lead is attacked by the etchants and only black voids will remain after etching. Therefore, micrographs to document the quantity and distribution of lead must be taken before etching. The color of pure lead is grey-blue. 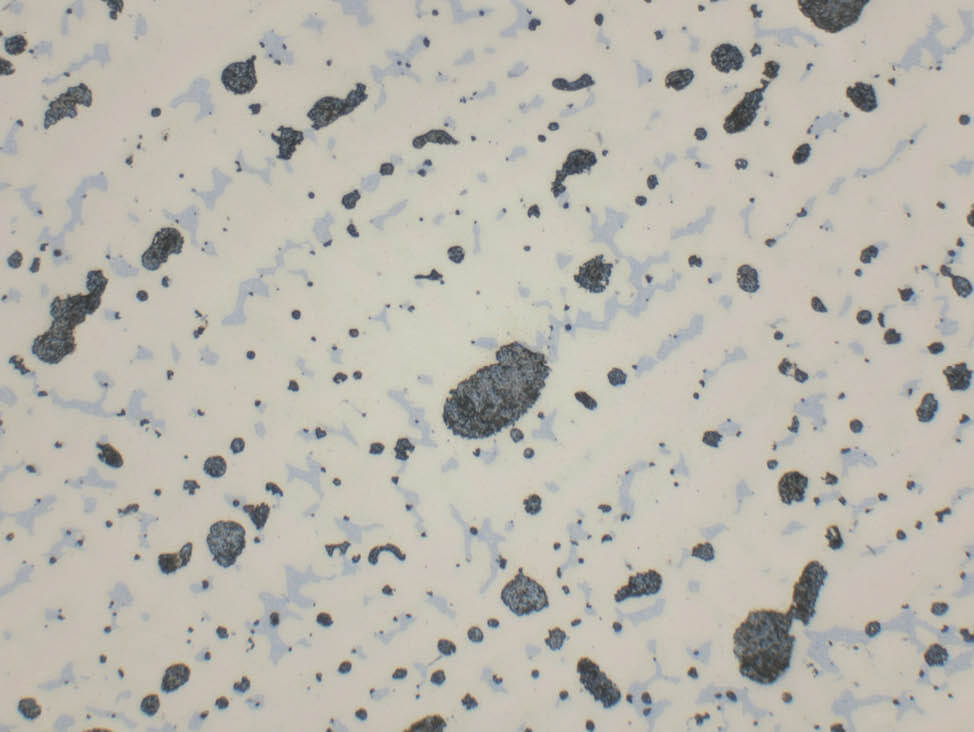
Fig. 13: Bronze cast (CuSn8Pb), unetched, showing large and small blue-grey lead inclusions, pale blue α-δ eutectoid discernable, 500x
Fig. 14: Same specimen as in Fig.13, color etched according to Klemm. The dendritic structure with light blue eutectoid and blue lead inclusions can be seen, but small lead inclusions cannot be clearly differentiated, 500x
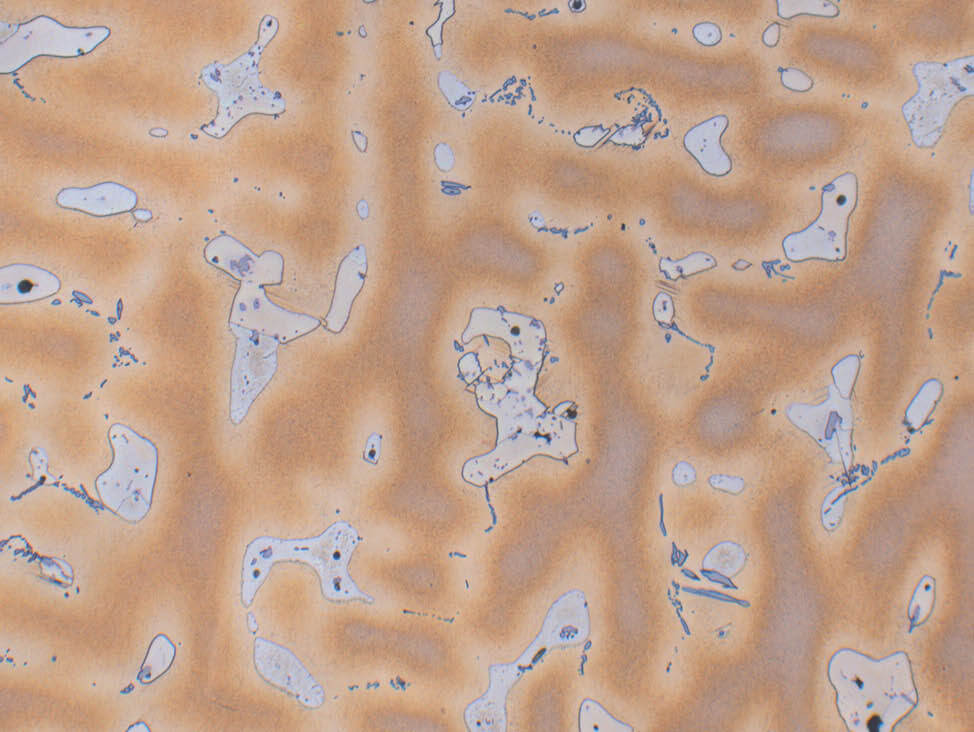
Fig. 15: Bronze cast (CuSn10), etched with iron (III) chloride, dendritic structure α-δ eutectoid, 200x
| Application | Etchant |
| Grain area etch for copper, brass and bronzes | 100 ml water. 10 g ammonium peroxydisulfate. Use fresh! |
| All types of copper | 100-120 ml water or ethanol. 20-50 ml hydrochloric acid. 5-10 g iron (III) chloride. (concentration variable) |
Grain boundaries Grain areas |
25 ml distilled water. 25 ml ammonia water. 5-25 ml hydrogen peroxide, 3% Less hydrogen peroxide More hydrogen peroxide |
| α-β brass | 120 ml water. 10 g copper (II) ammonium chloride. Add ammonia water until precipitate dissolves |
| Fast and good polish for pure copper | 100 ml water. 100 ml ethanol. 19 g iron (III) nitrate |
| Colour etch according to Klemm | 100 ml cold saturated sodium thiosulfate. 40 g potassium metabisulfit |
Download the application note to see the full preparation method
Summary
Due to its good formability, high electric and thermal conductivity and corrosion resistance, copper is found in a wide range of industries and applications, from electric engineering and electronics to the food and beverage industry.
The metallography of copper and its alloys is used in quality control, mainly to check purity and grain size determination. In addition, cast alloys are examined for general structure evaluation. As copper is soft and ductile, it is particularly prone to mechanical deformation. Therefore, the finest possible grit must be used for the first mechanical grinding step.
For successful preparation of copper and copper alloys with mechanical grinding, it is suggested that:
- Coarse abrasives during grinding are avoided
- Diamond polishing should be carried out on soft to medium-soft cloths
- Chemical-mechanical final polishing is essential to ensure a scratch-free surface
Get the full application note
Want to read the full application note on the metallographic preparation of copper and its alloys? Download it here
Get insight into other materials
If you would like to learn more about the materialography of other metals and materials, check out our materials pages.
For specific information about the metallographic preparation of Copper, contact our application specialists.