Metallographic preparation of titanium and titanium alloys

Titanium is a highly ductile metal, which makes it difficult to cut, grind and polish in preparation for microscopic observation. This guide outlines the most effective ways to prepare titanium and titanium alloys for metallographic examination, with accurate and reproducible results.
The main characteristics of titanium and titanium alloys
Titanium is mainly characterized by two features:
- High corrosion resistance: Its high affinity for oxygen gives it a thin, but dense, self-healing and stable oxide layer. This effectively protects the metal against incipient corrosion by a wide range of chemicals.
- High strength to weight ratio: This property is maintained even at higher temperatures.
Due to its chemical resistance and high strength to weight ratio, titanium and its alloys are widely used in the aerospace, chemical and medical industries, where safety and quality control are critical. Consequently, the materialographic preparation and analysis of titanium is essential.
The challenge when preparing titanium and titanium alloys for microscopic analysis is the high ductility, which makes the material prone to mechanical deformation. This must be taken into consideration during cutting, grinding and polishing.
Metallurgy and microstructure of titanium and titanium alloys
Metallography plays a key part in the quality control of titanium and its alloys, from monitoring the initial production process to assessing porosity on cast parts and controlling heat-treatment. It also plays an important part in research and development of titanium alloys and products.
Commercial titanium grades and alloys are divided into four groups:
- Unalloyed commercially pure titanium (CP)
- α and near α alloys, such as Ti-6Al-2Sn-4Zr-2Mo
- α-β alloys, such as Ti-6Al-4V
- β alloys, which have a high content of vanadium, chromium and molybdenum
At a temperature of 882°C, titanium undergoes an allotropic change from a low-temperature, close-packed hexagonal structure (α) to a body-centered cubic phase (β). This transformation makes it possible to create alloys with α, β or mixed α/β microstructures, and enables the use of heat treatment and thermo-mechanical treatment.
Consequently, a wide range of properties can be obtained from a relatively small number of alloy compositions. However, to ensure the desired combination of microstructure and properties, close control of the treatment process must be maintained. This makes metallography essential.
The relations between hot forming, heat treatment, microstructure and physical properties in the production of titanium and its alloys are very complex. A few examples of the most common types of titanium microstructures are shown below.

Fig. 1: Grain structure of commercially pure titanium, which has been mechanically deformed through bending. Twinning due to mechanical deformation is visible. Polarized light, 100x

Fig. 2: Structure of a forged α-β Ti-6Al-4V in the annealed condition. Etchant: Kroll’s reagent. 400x

Fig. 3: α-β Ti-6Al-4V with a white, brittle ‘α-case’ surface layer. Etchant: Weck’s reagent. Although hot forming processes are carried out under controlled atmosphere, titanium can absorb oxygen at lower temperatures, which results in a surface hardened zone, the α-case. This is a very brittle layer and can only be removed mechanically. 50x

Fig. 4: The β structure of the longitudinal section of a Ti-15V-3Al- 3Sn-3Cr alloy plate. This alloy is used in the aerospace industry because of its superior mechanical properties. Etchant: heat tinting. 50x
Preparation of titanium and titanium alloys: Cutting & mounting
Cutting titanium and titanium alloys
Because of its high ductility, machining or cutting titanium often produces long chips. For this reason, metallographic cutting with regular aluminum oxide cut-off wheels is highly ineffective, as it often leads to heat damage, as shown in figure 5.
The solution to this problem is to use silicon carbide cut-off wheels, which have been developed specifically for sectioning of titanium (e.g. 20SXX).
Cutting titanium generates a characteristic smell, which can be quite pronounced when cutting large pieces or quantities. In these cases, we recommend that you connect a fume extractor to the cut-off machine.

Fig. 5: Titanium can easily overheat during cutting, creating large burrs
Mounting titanium and titanium alloys
In primary production control labs, where mainly ingots, billets and slabs are analyzed, large samples of titanium are normally processed unmounted. However, smaller manufactured parts – such as wire and fasteners – should always be mounted. This simplifies preparation and ensures the best results with respect to accuracy and reproducibility.
For this, we recommend using hot-compression mounting with phenolic resin (MultiFast) or cold mounting with slow-curing epoxy (EpoFix).
Find out more
- Get more knowledge, expertise and insight in our cutting and mounting sections.
- See our range of cutting and mounting equipment.
- Get cutting consumables and accessories and mounting consumables and accessories.
Case story: Preparation time for titanium reduces from 60 to 11 minutes
Preparation of titanium and titanium alloys: Grinding & polishing
Because of its extreme ductility, titanium is prone to mechanical deformation and scratching during metallographic preparation. Diamond polishing should be avoided, especially with commercially pure titanium, as it introduces mechanical deformation to the surface in the form of scratches and smearing. Once introduced, this layer of deformation is difficult to remove. To avoid this issue, we recommend using a chemical-mechanical polish.

Fig. 6: Due to its ductility, titanium deforms and scratches easily. DIC, 50x
3-step method for grinding & polishing titanium and titanium alloys
This three-step automatic method is a proven procedure, which gives excellent, reproducible results for titanium and titanium alloys. (For more detail, see table 1.)
Step 1
Plane grinding with resin-bonded diamonds in a rigid disc, such as MD-Mezzo. (Note: When plane grinding pure titanium, a silicon carbide foil should be used, as shown in table 2).
Step 2
Fine grinding on a hard surface, such as MD-Largo or MD-Plan, using an abrasive 9 µm diamond suspension, such as DiaPro Allegro/Largo 9 or DiaPro Plan 9.
Step 3
Chemical-mechanical polishing with a mixture of colloidal silica (OP-S) and hydrogen peroxide (concentration between 10-30 %). Preparation time depends on the sample area and the titanium alloy. The larger the sample and the purer the titanium, the longer the final polishing step will take. Commercially pure titanium can take up to 45 minutes.
Continue polishing until the surface looks white in an optical microscope. Titanium and titanium alloys should normally look very clean after polishing, which means that any small black dots visible on the surface are most likely due to grinding deformation. These artefacts should be removed with further chemical-mechanical polishing. Once the polishing is complete, you should be able to see the structure in polarized light without etching.
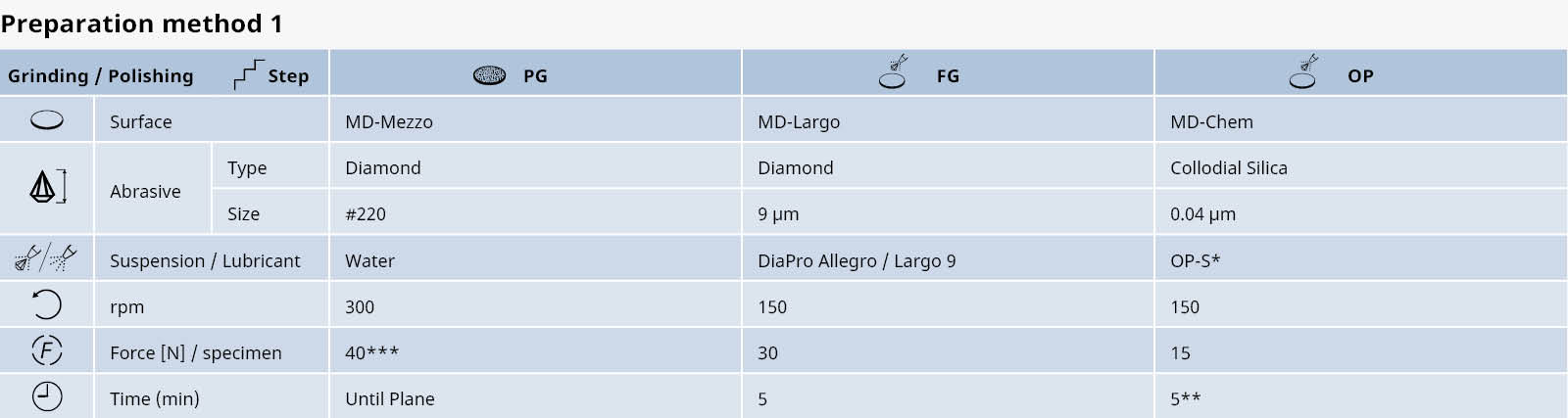
Table 1: Shows a general, automatic preparation method for titanium alloys with grade 5 or higher, unmounted samples, 30 mm dia. Please be aware that the polishing time can vary depending on the purity of the titanium and the area of the sample surface.
* Mix 70-90% OP-S with 10-30% H2O2 (of 30% conc).
** The polishing time depends on the sample area. Very large samples require more polishing time than small ones.
*** Decrease to 25 N to avoid pencil shapes in single sample preparation of mounted samples.
Note: during the last 20-30 seconds of the preparation step with OP-S, the rotating cloth is flushed with water. This will clean the samples, holder and cloth.
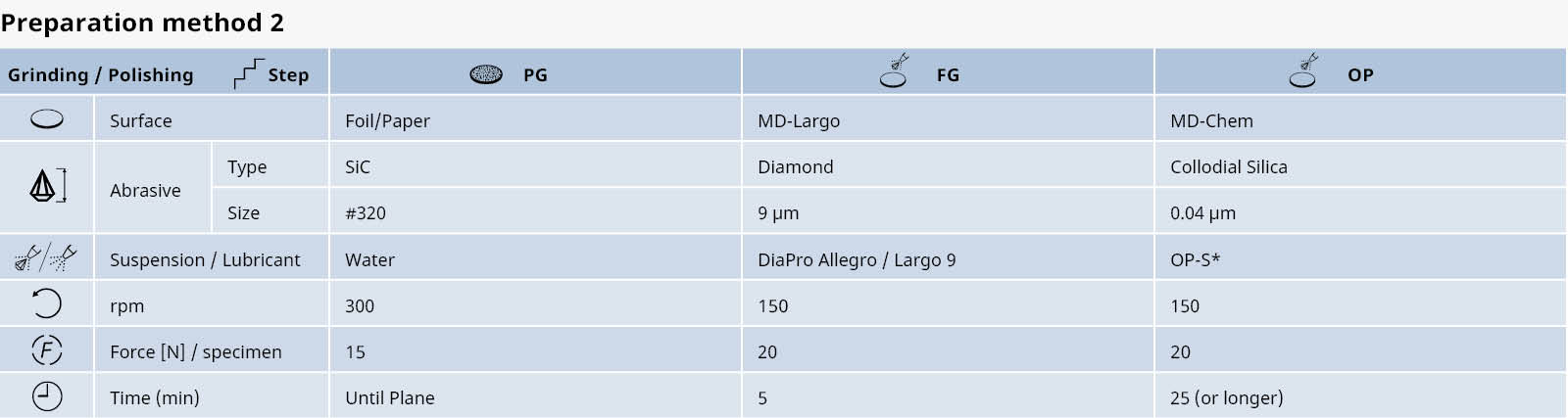
Table 2: Shows a general, automatic preparation method for pure titanium (grade 1-4) with unmounted samples, 30 mm dia. Please be aware that the polishing time can vary depending on the purity of the titanium and the area of the sample surface.
*80% OP-S + 10% H2O2 (30%) + 10% NH4OH (25%)

Fig. 7: Cross-section of a bar, commercially pure titanium, electrolytically polished. Polarized light. 100x
Chemical reagents for polishing titanium and titanium alloys
OP-S colloidal silica and hydrogen peroxide mixture
Unlike some other colloidal silica, OP-S was developed to accommodate chemical additions without transforming into a gel-like consistency. It is therefore well suited to polishing titanium and titanium alloys.
Hydrogen peroxide and titanium create a reaction product. During chemical-mechanical polishing, this is continuously removed from the sample’s surface by the silica suspension, leaving the surface free of mechanical deformation. When working with hydrogen peroxide, we recommend wearing rubber gloves.
Nitric and hydrofluoric acid mixture
Nitric and hydrofluoric acid mixtures can also be used for chemical-mechanical polishing of titanium. These reagents work very quickly. However, we do not recommend using them for polishing, because they are very corrosive and extensive safety precautions must be taken when handling these acids during the polishing procedure.

Fig. 8: Titanium after 3 µm diamond polish. Deformation and scratches are very difficult to remove
How to effectively wash your samples
When working with colloidal silica (OP-S), it is important to wet the cloth with water before starting to polish.
- Flush the rotating cloth with water for approximately 20-30 seconds before the machine stops to wash the OP-S off the samples, holder and cloth.
- Clean the samples again individually with neutral detergent and water.
- Dry the samples with ethanol and a strong stream of air. If you still see OP-S on the sample surface, repeat the cleaning process.
For a faster process with excellent and reproducible results, wash samples using automatic cleaning equipment, such as Lavamin.
Electrolytical polishing titanium and titanium alloys: a faster alternative to mechanical polishing
When you need fast results, electrolytical polishing is a useful option. α alloys, which have a homogenous structure, are particularly well suited for electrolytical polishing, but α-β alloys can also be polished electrolytically.
Electrolytical polishing of titanium and its alloys has clear advantages, including:
- Quick results (especially when it comes to pure titanium, which normally requires longer polishing times)
- Simple process
- Good reproducibility
- No mechanical deformation left on sample surfaces
The electrolytical polishing procedure requires a finely ground surface (with SiC #1200 or finer). After electrolytical polishing, the polished specimen can be examined in polarized light or etched chemically.
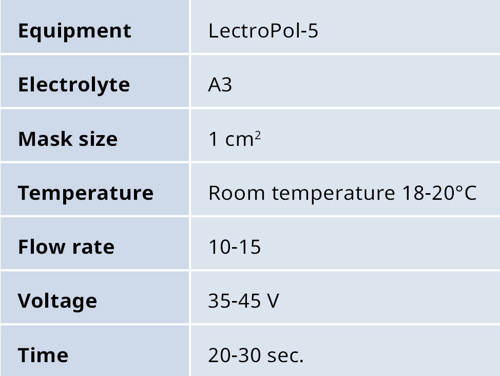
Table 3: Shows a general, electrolytic preparation method for pure titanium and titanium alloys.
Find out more
- Get more knowledge, expertise and insight in our grinding and polishing section.
- See our range of grinding and polishing machines and equipment.
- Get consumables and accessories for metallographic grinding and polishing.
Etching of titanium and titanium alloys
The surface of a well-polished titanium sample can be observed unetched in polarized light. Although the contrast is not very pronounced, this allows for a general check to see if the polish is sufficient.
The most common chemical etchant for titanium is Kroll’s reagent:
- 100 ml water
- 1-3 ml hydrofluoric acid
- 2-6 ml nitric acid
The concentration can vary depending on the alloy and can be adjusted individually. Kroll’s reagent colors the β phase dark brown.
Titanium can be color etched with Weck’s reagent:
- 100 ml water
- 5 g ammonium bifluoride
Summary
Titanium is a very ductile, low weight/high strength metal with excellent corrosion resistance and biocompatibility. It is used extensively in the aerospace, chemical and medical industries, where safety and quality control are critical.
Titanium’s ductility requires a specific metallographic preparation, using special cut-off wheels for sectioning and a chemical-mechanical polish with a mixture of hydrogen peroxide and colloidal silica. This polishing method, carried out with automatic equipment, gives consistently excellent and reproducible results.
Get insight into other materials
If you would like to learn more about the materialography of other metals and materials, check out our materials page.
All images by Marcello Manca, application specialist, Denmark
For specific information about the metallographic preparation of Titanium, contact our application specialists.
